Mac Olania ODT5 5 mg
For the use of a Neurologist or Registered Medical Practitioner or Hospital or a Laboratory only
OLANIA ODT
COMPOSITION
OLANIA ODT
Each uncoated orodispersible tablet contains:
Olanzapine Ph.Eur. ……………………… 5 mg / 10 mg
Excipients …………………………….….…. q.s.
DESCRIPTION
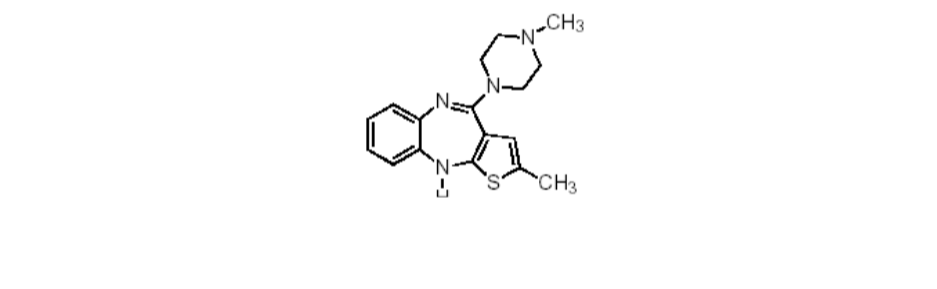
Olanzapine is a psychotropic agent that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b][1,5] benzodiazepine. It has a molecular weight of 312.44. The molecular formula is C17H20N4S and the chemical structure is:
Olanzapine is a yellow crystalline solid, which is practically insoluble in water.
Olanzapine or dispersible tablets begins disintegrating in the mouth within seconds, allowing its contents to be subsequently swallowed with or without liquid.
CLINICAL PHARMACOLOGY
-
Pharmacodynamics Olanzapine is an antipsychotic agent that demonstrates a broad pharmacologic profile across a number of receptor systems. Olanzapine exhibited a range of receptor affinities (Ki; < 100 nM) for serotonin 5 HT2A/2C, 5 HT3, 5 HT6; dopamine D 1 , D 2 , D 3 , D 4 , D 5 ; cholinergic muscarinic receptors. It is reported that olanzapine reduced a conditioned avoidance response, a test indicative of antipsychotic activity, at doses below those producing catalepsy, an effect indicative of motor side-effects. Unlike some other antipsychotic agents, olanzapine increases responding in an “anxiolytic” test.Olanzapine produced a higher 5-HT 2 A than dopamine D 2 receptor occupancy. In addition, olanzapine-responsive patients had lower striatal D 2 occupancy than some other antipsychotic and risperidone-responsive patients, while being comparable to clozapine-responsive patients.
It is reported that olanzapine was associated with statistically significantly greater improvements in negative as well as positive symptoms.
It is reported that olanzapine demonstrated statistically significant superiority over placebo on the primary endpoint of bipolar recurrence. Olanzapine also reported a statistically significant advantage over placebo in terms of preventing either recurrence into mania or recurrence into depression. -
Pharmacokinetics Olanzapine is well absorbed after oral administration reaching peak plasma concentrations within 5 to 8 hours. The absorption is not affected by food. Olanzapine is metabolized in the liver by conjugative and oxidative pathways. The major circulating metabolite is the 10-N-glucuronide which does not pass the blood brain barrier. Cytochromes P450- CYP1A2 and P450-CYP2D6 contribute to the formation of the N- desmethyl and 2-hydroxymethyl metabolites. It is reported that both exhibited significantly less in vivo pharmacological activity than olanzapine. The predominant pharmacologic activity is from the parent olanzapine. After oral administration, the mean terminal elimination half-life of olanzapine in healthy subjects varied on the basis of age and gender.It is reported that in healthy elderly (65 and over) versus non-elderly subjects, the mean elimination half-life was prolonged (51.8 versus 33.8 hr) and the clearance was reduced (17.5 versus 18.2 l/hr). The pharmacokinetic variability reported in the elderly is within the range for the non-elderly. It is reported that in 44 patients with schizophrenia >65 years of age, dosing from 5 to 20 mg/day was not associated with any distinguishing profile of adverse events. The plasma clearance of olanzapine is lower in elderly versus young subjects, in females versus males, and in non-smokers versus smokers. However, the magnitude of the impact of age, gender, or smoking on olanzapine clearance and half-life is small in comparison to the overall variability between individuals.
Adolescents (ages 13 to 17 years) – The pharmacokinetics of olanzapine are similar between adolescents and adults. It is reported that the average olanzapine exposure was approximately 27% higher in adolescents. Demographic differences between the adolescents and adults include a lower average body weight and fewer adolescents were smokers. Such factors likely contribute to the higher average exposure reported in adolescents.
The plasma protein binding of olanzapine was about 93 % over the concentration range of about 7 to about 1000 ng/mL. Olanzapine is bound predominantly to albumin and α 1 –acid- glycoprotein.
INDICATIONS
Olanzapine is indicated for the treatment of schizophrenia. Olanzapine is effective in maintaining the clinical improvement during continuing therapy in patients who have shown initial treatment response. Olanzapine is indicated for short-term treatment of acute manic episode associated with Bipolar I Disorder. Olanzapine is indicated for preventing recurrence of manic, mixed or depressive episodes in Bipolar I Disorder.
CONTRAINDICATION
Hypersensitivity to olanzapine or any of the excipients. Patients with known risk for narrow- angle glaucoma.
WARNINGS AND PRECAUTIONS
During antipsychotic treatment, improvement in the patient’s clinical condition may take several days to some weeks. Patients should be closely monitored during this period.
-
Dementia-related psychosis and/or behavioral disturbances Olanzapine is not approved for the treatment of dementia-related psychosis and/or behavioural disturbances and is not recommended for use in this particular group of patients because of an increase in mortality and the risk of cerebrovascular accident. There was a 2-fold increase in the incidence of death reported in olanzapine-treated patients. The higher incidence of death was not associated with olanzapine dose (mean daily dose 4.4 mg) or duration of treatment. Risk factors that may predispose this patient population to increased mortality include age > 65 years, dysphagia, sedation, malnutrition and dehydration, pulmonary conditions (e.g., pneumonia, with or without aspiration), or concomitant use of benzodiazepines. However, the incidence of death was higher in olanzapine-treated independent of these risk factors.Cerebrovascular adverse events (CVAE e.g., stroke, transient ischaemic attack), including fatalities, were also reported with use of olanzapine. All olanzapine patients who experienced a cerebrovascular event had pre-existing risk factors. Age> 75 years and vascular/mixed type dementia were reported as risk factors for CVAE in association with olanzapine treatment.
-
Parkinson’s disease The use of olanzapine in the treatment of dopamine agonist associated psychosis in patients with Parkinson’s disease is not recommended. Worsening of Parkinsonian symptomatology and hallucinations were reported with use of olanzapine.
-
Neuroleptic Malignant Syndrome (NMS)NMS is a potentially life-threatening condition associated with antipsychotic medicinal products. Rare cases reported as NMS have also been reported in association with olanzapine. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure. If a patient develops signs and symptoms indicative of NMS, or presents with unexplained high fever without additional clinical manifestations of NMS, all antipsychotic medicines, including olanzapine must be discontinued.
-
Hyperglycemia and diabetes mellitusHyperglycaemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycaemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of treatment-emergent hyperglycaemia- related adverse events in patients treated with the atypical antipsychotics. Precise risk estimates for hyperglycaemia-related adverse events in patients treated with atypical antipsychotics are not available.Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycaemia including polydipsia, polyuria; polyphagia, and weakness. Patients who develop symptoms of hyperglycaemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of antidiabetic treatment despite discontinuation of the suspect drug.
-
Lipid alterations Undesirable alterations in lipids have been reported in olanzapine-treated patients. Lipid alterations should be managed as clinically appropriate, particularly in dyslipidemic patients and in patients with risk factors for the development of lipids disorders. Patients treated with any antipsychotic agents, including olanzapine, should be monitored regularly for lipids in accordance with utilised antipsychotic guidelines, e.g., at baseline, 12 weeks after starting olanzapine treatment and every 5 years thereafter.
-
Anticholinergic activity Olanzapine had anticholinergic activity. As clinical experience with olanzapine in patients with concomitant illness is limited, caution is advised when prescribing for patients with prostatic hypertrophy, or paralytic ileus and related conditions.
-
Hepatic function Transient, asymptomatic elevations of hepatic aminotransferases, ALT, AST have been reported commonly, especially in early treatment. Caution should be exercised and follow-up organised in patients with elevated ALT and/or AST, in patients with signs and symptoms of hepatic impairment, in patients with pre-existing conditions associated with limited hepatic functional reserve, and in patients who are being treated with potentially hepatotoxic medicines. In cases where hepatitis (including hepatocellular, cholestatic or mixed liver injury) has been diagnosed, olanzapine treatment should be discontinued.
-
Neutropenia Caution should be exercised in patients with low leucocyte and/or neutrophil counts for any reason, in patients receiving medicines known to cause neutropenia, in patients with a history of drug- induced bone marrow depression/toxicity, in patients with bone marrow depression caused by concomitant illness, radiation therapy or chemotherapy and in patients with hypereosinophilic conditions or with myeloproliferative disease. Neutropenia has been reported commonly when olanzapine and valproate are used concomitantly.
-
Discontinuation of treatment Acute symptoms such as sweating, insomnia, tremor, anxiety, nausea, or vomiting have been reported when olanzapine is stopped abruptly.
-
QT intervalAs with other antipsychotics, caution should be exercised when olanzapine is prescribed with medicines known to increase QTc interval, especially in the elderly, in patients with congenital long QT syndrome, congestive heart failure, heart hypertrophy, hypokalaemia or hypomagnesaemia.
-
Thromboembolism Temporal association of olanzapine treatment and venous thromboembolism has been reported with use of olanzapine. A causal relationship between the occurrence of venous thromboembolism and treatment with olanzapine has not been reported. However, since patients with schizophrenia often present with acquired risk factors for venous thromboembolism, all possible risk factors of VTE e.g., immobilisation of patients, should be identified and preventive measures undertaken.
-
General CNS activityGiven the primary CNS effects of olanzapine, caution should be used when it is taken in combination with other centrally acting medicines and alcohol. Olanzapine may antagonise the effects of direct and indirect dopamine agonists.
-
SeizuresOlanzapine should be used cautiously in patients who have a history of seizures or are subject to factors which may lower the seizure threshold. Seizures have been reported to occur rarely in patients when treated with olanzapine. In most of these cases, a history of seizures or risk factors for seizures were reported.
-
Tardive dyskinesiaOlanzapine was reported a lower incidence of treatment-emergent dyskinesia. However, the risk of tardive dyskinesia increases with long-term exposure, and therefore if signs or symptoms of tardive dyskinesia appear in a patient on olanzapine, a dose reduction or discontinuation should be considered. These symptoms can temporally deteriorate or even arise after discontinuation of treatment.
-
Postural hypotension Postural hypotension was reported in the elderly in olanzapine. It is recommended that blood pressure is measured periodically in patients over 65 years.
-
Sudden cardiac deathThe event of sudden cardiac death has been reported in patients with olanzapine. The risk of presumed sudden cardiac death in patients treated with olanzapine was reported approximately twice the risk in patients not using antipsychotics. The risk of olanzapine was reported comparable to the risk of atypical antipsychotics.
-
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported with olanzapine exposure. DRESS consists of a combination of three or more of the following: cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, lymphadenopathy and one or more systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and pericarditis. Discontinue olanzapine if DRESS is suspected.
-
Paediatric population Various adverse reactions, including weight gain, changes in metabolic parameters and increases in prolactin levels were reported in patients aged 13-17. Long-term outcomes associated with these events have not been reported and remain unknown.
-
Phenylalanine This formulation contains aspartame, which is a source of phenylalanine. May be harmful for people with phenylketonuria.
-
Mannitol This formulation contains mannitol.
DRUG INTERACTION
Caution should be exercised in patients who receive medicinal products that can cause central nervous system depression.
Potential interactions affecting olanzapine: Since olanzapine is metabolized by CYP1A2, substances that can specifically induce or inhibit this isoenzyme may affect the pharmacokinetics of olanzapine.
Induction of CYP1A2: The metabolism of olanzapine may be induced by smoking and carbamazepine, which may lead to reduced olanzapine concentrations. Only slight to moderate increase in olanzapine clearance has been observed. The clinical consequences are likely to be limited, but clinical monitoring is recommended and an increase of olanzapine dose may be considered if necessary.
Inhibition of CYP1A2: Fluvoxamine a specific CYP1A2 inhibitor has been shown to significantly inhibit the metabolism of olanzapine. The mean increase in olanzapine Cmax following fluvoxamine was 54 % in female nonsmokers and 77 % male smokers. The mean increase in olanzapine AUC was 52 % and 108 % respectively. A lower starting dose of olanzapine should be considered in patients who are using fluvoxamine or any other CYP1A2 inhibitors, such as ciprofloxacin or ketoconazole. A decrease in the dose of olanzapine should be considered if treatment with an inhibitor of CYP1A2 is initiated.
Decreased bioavailability: Activated charcoal reduces the bioavailability of oral olanzapine by 50 to 60% and should be taken at least 2 hours before or after olanzapine, Fluoxetine (a CYP2D6 inhibitor), single doses of antacid (aluminum, magnesium) or cimetidine have not been found to significantly affect the pharmacokinetics of olanzapine.
Potential for olanzapine to affect other medicinal products: Olanzapine may antagonize the effects of direct and indirect dopamine agonists.
Olanzapine does not inhibit the main CYP450 isoenzymes in vitro (e.g. 1A2, 2D6, 2C9, 2C19.3A4). Thus no particular interaction is expected as verified through in vivo studies where no inhibition of metabolism of the following active substances was found: tricyclic antidepressant (representing mostly CYP2D6 pathway), warfarin (CYP2C9), theophyline (CYP1A2) or diazepam (CYP3A4 and 2C19), Olanzapine showed no interaction when co- administered with lithium or biperiden.
Therapeutic monitoring of valproate plasma levels did not indicate that valproate dosage adjustment is required after the introduction of concomitant olanzapine.
ADVERSE EFFECTS
- Adults
Blood and the lymphatic system disorders Eosinophilia, Leucopenia, Neutropenia, Thrombocytopenia
Immune system disorders Allergic reaction
Metabolism and nutrition disorders Weight gain, Elevated cholesterol levels, Elevated glucose levels, Elevated triglyceride levels, Glucosuria, Increased appetite, Development or exacerbation of diabetes occasionally associated with ketoacidosis or coma, including some fatal cases, Hypothermia
Nervous system disorders Somnolence, Dizziness, Akathisia, Parkinsonism, Dyskinesia, Seizures where in most cases a history of seizures or risk factors for seizures were reported, Neuroleptic malignant syndrome, Dystonia (including oculogyration), Tardive dyskinesia, Discontinuation symptoms, Restless legs syndrome
Cardiac disorders Bradycardia, QTc prolongation, entricular tachycardia/fibrillation, sudden death
Vascular disorders Orthostatic hypotension, Thromboembolism (including pulmonary embolism and deep vein thrombosis)
Gastrointestinal disorders Mild, transient anticholinergic effects including constipation and dry mouth, Pancreatitis
Hepato-biliary disorders Transient, asymptomatic elevations of hepatic aminotransferases (ALT, AST), especially in early treatment, Hepatitis (including hepatocellular, cholestatic or mixed liver injury)
Skin and subcutaneous tissue disorders Rash, Photosensitivity reaction, Alopecia
Very rare: Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS).Musculoskeletal and connective tissue disorders Rhabdomyolysis
Renal and urinary disorders Urinary incontinence, urinary retention, Urinary hesitation
Pregnancy, puerperium and perinatal conditions Drug withdrawal syndrome neonatal
Reproductive system and breast disorders Erectile dysfunction in males, Decreased libido in males and females, Amenorrhea, Breast enlargement, Galactorrhea in females, Gynaecomastia/breast enlargement in males, Priapism
General disorders and administration site conditions Asthenia, Fatigue, Oedema
Investigations Elevated plasma prolactin levels, High creatine phosphokinase, Increased total bilirubin, Increased alkaline phosphatase
Respiratory, Thoracic and Mediastinal Disorders: Sleep apnoea*
*Atypical antipsychotic drugs, such as Olanzapine, have been associated with cases of sleep apnoea, with or without concomitant weight gain. In patients who have a history of or are at risk for sleep apnoea, OLANIA ODT Tablets should be prescribed with caution.
-
Long-term exposure (at least 48 weeks)The proportion of patients who had adverse, clinically significant changes in weight gain, glucose, total/LDL/HDL cholesterol or triglycerides reported increased over time. In adult patients who completed 9-12 months of therapy, the rate of increase in mean blood glucose slowed after approximately 6 months.
-
Additional information on special populationsIn elderly patients with dementia, olanzapine treatment was associated with a higher incidence of death and cerebrovascular adverse reactions. Very common adverse reactions reported with the use of olanzapine in this patient group were abnormal gait and falls. Pneumonia, increased body temperature, lethargy, erythema, visual hallucinations and urinary incontinence were reported commonly. Patients with drug-induced (dopamine agonist) psychosis associated with Parkinson’s disease, worsening of Parkinsonian symptomatology and hallucinations were reported very commonly.In patients with bipolar mania, valproate combination therapy with olanzapine reported an incidence of neutropenia; a potential contributing factor could be high plasma valproate levels.
Olanzapine administered with lithium or valproate reported in increased levels of tremor, dry mouth, increased appetite, and weight gain. Speech disorder was also reported commonly. During treatment with olanzapine in combination with lithium or divalproex, an increase body weight occurred in patients during acute treatment (up to 6 weeks). Long-term olanzapine treatment (up to 12 months) for recurrence prevention in patients with bipolar disorder was reported with an increase body weight.
-
Paediatric population Olanzapine is not indicated for the treatment of children and adolescent patients below 18 years.
| Metabolism and nutrition disorders
Weight gain, elevated triglyceride levels 10 , increased appetite, Elevated cholesterol levels |
| Nervous system disorders
Sedation (including: hypersomnia, lethargy, somnolence). |
| Gastrointestinal disorders
Dry mouth. |
| Hepato-biliary disorders
Elevations of hepatic aminotransferases (ALT/AST) |
| Investigations
Decreased total bilirubin, increased GGT, elevated plasma prolactin levels. |
OVERDOSAGE
Very common symptoms in overdose include tachycardia, agitation, aggressiveness, dysarthria, various extrapyramidal symptoms, and reduced level of consciousness ranging from sedation to coma. Other medically significant sequelae of overdose include delirium, convulsion, coma, possible neuroleptic malignant syndrome, respiratory depression, aspiration, hypertension or hypotension, cardiac arrhythmias and cardiopulmonary arrest. Fatal outcomes have been reported for acute overdoses as low as 450mg but survival has also been reported following acute overdose of 1,500mg.
-
Management of Overdose There is no specific antidote for olanzapine. Induction of emesis is not recommended. Standard procedures for management of overdose may be indicated (i.e. gastric lavage, administration of activated charcoal). The concomitant administration of activated charcoal was shown to reduce the oral bioavailability of olanzapine by 50 to 60%. Symptomatic treatment and monitoring of vital organ function should be instituted according to clinical presentation, including treatment of hypotension and circulatory collapse and support of respiratory function. Do not use epinephrine, dopamine, or other sympathomimetic agents with beta-agonist activity since beta stimulation may worsen hypotension. Cardiovascular monitoring is necessary to detect possible arrhythmias. Close medical supervision and monitoring should continue until the patient recovers.
DOSAGES AND ADMINISTRATION
- AdultsSchizophrenia:
The recommended starting dose for olanzapine is 10 mg/day. Manic episode: The starting dose is 15 mg as a single daily dose in monotherapy or 10 mg daily in combination therapy.Preventing recurrence in bipolar disorder: The recommended starting dose is 10 mg/day. For patients who have been receiving olanzapine for treatment of manic episode, continue therapy for preventing recurrence at the same dose. If a new manic, mixed, or depressive episode occurs, olanzapine treatment should be continued (with dose optimisation as needed), with supplementary therapy to treat mood symptoms, as clinically indicated.
During treatment for schizophrenia, manic episode, and recurrence prevention in bipolar disorder, daily dosage may subsequently be adjusted on the basis of individual clinical status within the range 5-20 mg/day. An increase to a dose greater than the recommended starting dose is advised only after appropriate clinical reassessment and should generally occur at intervals of not less than 24 hours. Olanzapine can be given without regard for meals, as absorption is not affected by food. Gradual tapering of the dose should be considered when discontinuing olanzapine.
Olanzapine or dispersible tablet should be placed in the mouth, where it will rapidly disperse in saliva, so it can be easily swallowed. Removal of the intact or dispersible tablet from the mouth is difficult. Since the or dispersible tablet is fragile, it should be taken immediately on opening the blister. Alternatively, it may be dispersed in a full glass of water or other suitable beverage (orange juice, apple juice, milk, or coffee) immediately before administration. Olanzapine or dispersible tablet has the same dosage and frequency of administration as olanzapine coated tablets. Olanzapine or dispersible tablets may be used as an alternative to olanzapine coated tablets.
-
Paediatric population Olanzapine is not recommended for use in children and adolescents below 18 years. A greater magnitude of weight gain, lipid and prolactin alterations has been reported adolescent patients than of adult patients.
-
Elderly patientsA lower starting dose (5 mg/day) is not routinely indicated but should be considered for those 65 and over when clinical factors warrant.
-
Patients with renal and/or hepatic impairment lower starting dose (5 mg) should be considered for such patients. In cases of moderate hepatic insufficiency (cirrhosis, Child-Pugh class A or B), the starting dose should be 5 mg and only increased with caution.
-
Generate the starting dose and dose range need not be routinely altered for female patients relative to male patients.
-
Smokers the starting dose and dose range need not be routinely altered for non-smokers relative to smokers.When more than one factor is present which might result in slower metabolism (female gender, geriatric age, non-smoking status), consideration should be given to decreasing the starting dose. Dose escalation, when indicated, should be conservative in such patients.
-
Method of administration Olanzapine orodispersible tablet should be placed in the mouth, where it will rapidly disperse in saliva, so it can be easily swallowed. Removal of the intact or dispersible tablet from the mouth is difficult. Since the orodispersible tablet is fragile, it should be taken immediately on opening the blister. Alternatively, it may be dispersed in a full glass of water or other suitable beverage (orange juice, apple juice, milk or coffee) immediately before administration.
-
USE IN PREGNANCY, NURSING MOTHER AND FERTILITY
-
Pregnancy Olanzapine falls under Pregnancy Category C. Olanzapine should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus.
-
Breast-feeding It is recommended that women receiving olanzapine should not breast-feed.
-
Fertility Rodent studies suggest that olanzapine may produce a delay in ovulation.
PRESENTATION:
OLANIA ODT Tablets are packed in Alu-Alu blister of 10 tablets. Such blisters containing 10 Tablets are packed into boxes of 10’s, 30’s, 60’s and 100’s.



